April 23, 2026
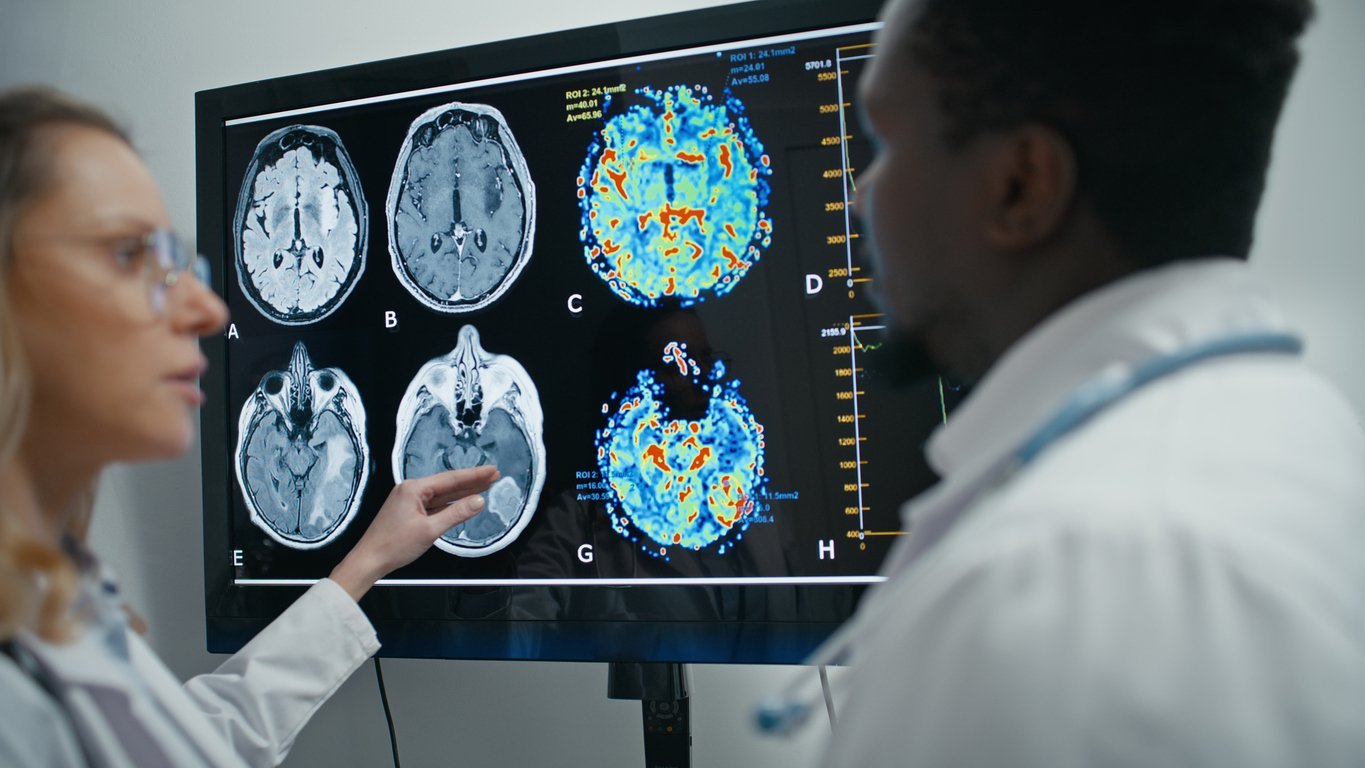
Curtana Pharmaceuticals Receives FDA Rare Pediatric Disease Designation for CT-179 in Pediatric High-Grade Glioma
Designation for first-in-class OLIG2 inhibitor highlights the urgent need for new therapies in aggressive pediatric brain cancers and qualifies Curtana for a potential Priority Review Voucher upon FDA approval. Austin, TX – April 23, 2026 – Curtana Pharmaceuticals, a clinical-stage biotechnology company focused on improving outcomes for brain cancer patients, today announced that the U.S.…